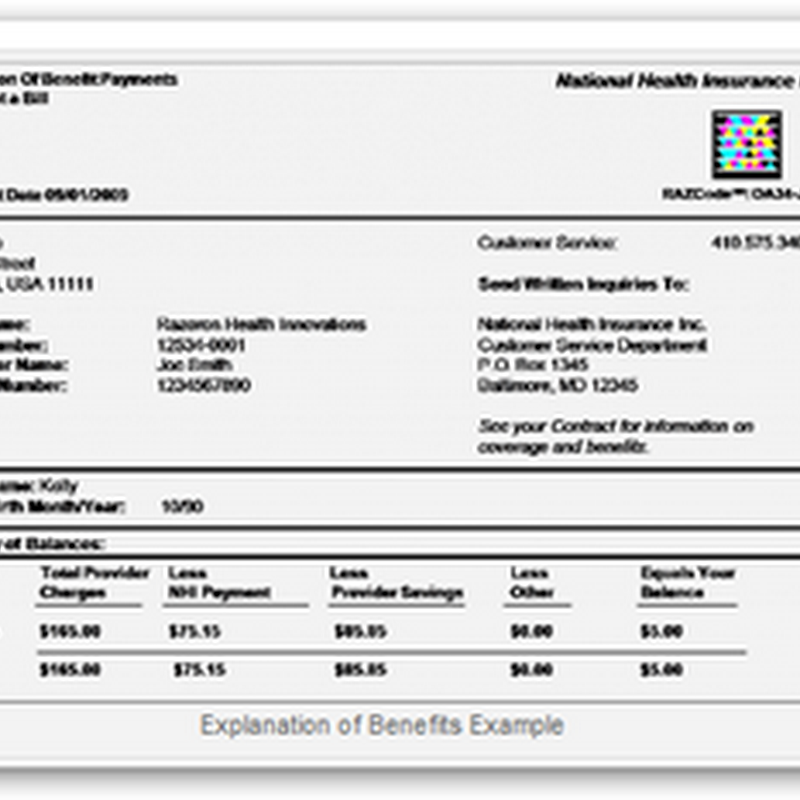
Private Equity Firm Golder Rauner LLC (Devicor Unit) Makes Offer for J and J Ethicon Breast Care Business
Private Equity Firm Golder Rauner LLC (Devicor Unit) Makes Offer for J and J Ethicon Breast Care Business
Below is a statement from the Devicor website, so it appears their business acquiring and growing healthcare companies. The article also m...