Back in June of this year I reported on the FDA approval of their hand held device and here we are 6 months later and gone. The company had been around since 1984. 300 more people will be looking for work in Minnesota. They also had strong financial backers throughout but ran out of money. Hospitals are looking to medical device companies for relief, and perhaps some can help but not companies that are borderline on the verge of running out of money. This company was in a market that is projected to hit over 2 billion with medical devices that report data, implanted and otherwise.
Hospitals are looking to medical device companies for relief, and perhaps some can help but not companies that are borderline on the verge of running out of money. This company was in a market that is projected to hit over 2 billion with medical devices that report data, implanted and otherwise.
Transoma Medical Receives FDA Marketing Clearance – ECGs on Mobile Handheld Device from Implanted Device
This brings me back around once more to implantation and marketing. Many companies have a short revenue life line, thus they need to sell and market products immediately. What we are beginning to see is healthcare devices that report data are literally being almost tossed out there to grab some dollars. Some companies may not be doing that but will they survive?
It’s not really perhaps survival of the best product, it seems to be more of survival of the best “cash cow” these days. Again I see this many times with the research and marketing I do, it’s a rush to keep the doors open, and thus the marketing information we get may not always be clear, understandable, and furthermore correct. Look at the recent problems Stryker had with marketing and the indictments, and one other company Norian who did illegal clinical trials and people died. In this post we are talking devices, not drugs. It’s in the air, so patient and doctor beware. BD
Here’s some information from my prior post earlier this year – implanted device that sends data – keep your eyes open.
“Information is sent through frequent ECG samples to Manage Complex and often Changing Arrhythmias with patients to a cardio center for technicians to analyze. From here physicians are alerted for any significant events. One nice move is a physician’s portal, but we need to go the next level up and have the information brought to a Personal Health Record, so perhaps this is a new potential vendor for Microsoft.
Cardio implants are now being capable of sending SMS, faxes and emails themselves too. With this device, the patients too can see the information and thus the need to include in a PHR. The procedures is minimally invasive, since the device is not providing a function for the heart, but rather creating a ECG electronic report card. With the FDA marketing clearance, we could be seeing this device recommended by physicians soon. It does make you wonder exactly how much time will transpire before we all have some type of device someday.” 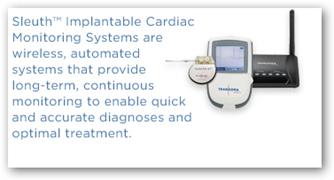
ARDEN HILLS, Minnesota–Transoma Medical Inc. has become the latest medical device start-up to shut its doors.
The Arden Hills-based company, which was developing a device to remotely monitor the electrical activity of a patient’s heart, could not raise additional money from investors, sources said. As a result, Transoma closed its human applications operations and will return to its original animal business.
The company employed more than 300 people. In 2007, Transoma generated $37.2 million in revenue.
It’s a stunning fall for Transoma, which had already raised millions of dollars from investors like Affinity Capital Partners in Minneapolis and had once filed for a $75 million initial public offering only to withdraw it last year because of poor market conditions. The company had extensively developed and tested its Sleuth device, which allowed doctors to monitor and collect data on patients who suffer from sudden fainting spells.
Founded in 1984 as Data Sciences International, Transoma’s expertise was pharmaceutical research; it developed implantable wireless devices to monitor the effects of drugs on animals. The company decided to focus on patients who had chronic heart-related conditions such as syncope and refractory epilepsy, who could benefit from long-term monitoring — a potential $2.1 billion U.S. market.
But a weak economy, which scared off investors, and a poor IPO environment worked against Transoma. The company’s demise does not bode well for Minnesota medical start-ups, said Ross Meisner, managing director of Dymedex Consulting in Vandais Heights. Transoma boasted prominent investors, won regulatory approval and generated real sales but still failed, he said.
“Transoma is a good example of how challenging the market is right now,” Meisner said.
Minnesota’s Transoma Medical shuts down : MedCity News



0 comments :
Post a Comment