New trial to begin with the device that is a miniaturized implantable heart pump..for  patients with advanced heart failure...clinical trials already in place in Europe and Australia...the endpoint for survival is 180 days...patients do not need to be lsited for a transplant to be considered a success...the trial again is for patients with advanced issues and the patients will no be required to stay at the hospital facility...BD
patients with advanced heart failure...clinical trials already in place in Europe and Australia...the endpoint for survival is 180 days...patients do not need to be lsited for a transplant to be considered a success...the trial again is for patients with advanced issues and the patients will no be required to stay at the hospital facility...BD
HeartWare Limited (ASX: HTW) announced that it had received conditional approval from the United States Food and Drug Administration (FDA) of an Investigational Device Exemption (IDE) for its lead product, the HeartWare(R) Left Ventricular Assist System (LVAS).
"The HVADTM pump is a small centrifugal blood pump capable of generating up to 10 litres per minute of forward flow. The pump is designed to draw blood from the apex of the left ventricle and to propel it through an outflow graft connected to the patient's ascending aorta.
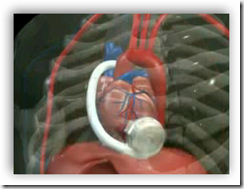
With a displaced volume of only 45cc, the HVADTM pump is designed to be implanted in the pericardial space, directly adjacent to the heart and above the diaphragm. Implantation above the diaphragm is expected to lead to short surgery time and quick recovery."



0 comments :
Post a Comment