This is an interesting medical device from Uroplasty called the “Urgent PC” and requires a few treatments and they do a demonstration on the video below. 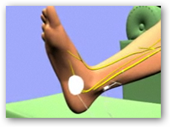 Patients with pacemakers or implanted defibrillators though are out of the picture for obvious reasons of potential interference.
Patients with pacemakers or implanted defibrillators though are out of the picture for obvious reasons of potential interference.
I addition the site has a page on how to code for reimbursement. It is broken down by areas in each state. The American Urological Association (AUA) and the American Medical Association (AMA) have recently suggested that PTNS should be reported with CPT code 64999.
This really looks like a great idea and has been approved by the FDA since 2006. The only other device that treats incontinence is made by Medtronic and is implanted where this one is not. The initial treatment is a 30 minute session for 12 weeks, and afterwards one treatment every 21 days.
From the Website:
“The Urgent PC Neuromodulation System is a combination of a stimulator and a lead set. The stimulator generates a specific kind of electrical impulse that is delivered to the patient through the lead set. Using a needle electrode placed near the ankle as an entry point, the stimulator’s impulses travel along the tibial 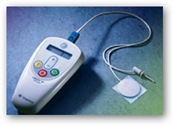 nerve to the nerves in the spine that control pelvic floor function.
nerve to the nerves in the spine that control pelvic floor function.
How does neuromodulation work?
Bladder function is regulated by a group of nerves at the base of the spine called the sacral nerve plexus. By stimulating these nerves through gentle electrical impulses (neuromodulation), your bladder activity can be changed.
The Urgent PC System provides neuromodulation in a low-risk, office procedure by indirectly stimulating the nerves responsible for bladder function using a nerve in your lower leg. “
The company is hoping to get more insurers to cover the procedure as well as Medicare. It certainly looks simple enough and you can just sit down and read while undergoing the therapy. BD
Earlier this week, the Minnetonka company published positive results of a study that treated 220 patients suffering from incontinence — specifically those who were dealing with frequent urges — with electrical nerve stimulation, or neurostimulation. The procedure was pegged against a sham procedure, much as an experimental drug is tested against a placebo. Results showed that 58.3 percent of patients who used Uroplasty’s neurostimulation treatment reported improvements in symptoms, compared with 21.9 percent of the patients who were given the sham procedure.
The results of the study will be published in the April edition of the Journal of Urology. This latest study follows two other studies also published in the same journal. One study showed the efficacy of the company’s Urgent PC system compared to the most often prescribed drug for incontinence, Detrol. Through Urgent PC, a needle electrode is inserted into the ankle of a patient and a pad electrode is placed on the foot hooked to the stimulator. When the stimulator is turned on, an electric current passes through the tibial nerve to the sacral plexus, which controls bladder and pelvic floor function.
Local company hopes to challenge Medtronic incontinence product - Finance and Commerce




0 comments :
Post a Comment