There are just a little over 5000 units to be recalled and this is one more case to where the packaging units as well as a synchronized data base at the FDA could have been used to find the recalled products. The concern is that the Introducer  device could become detached during use. The actual recall date was June 24, 2010 and the company has sent out notices and is relying on sales reps to follow up with facilities who purchased the units.
device could become detached during use. The actual recall date was June 24, 2010 and the company has sent out notices and is relying on sales reps to follow up with facilities who purchased the units.
Microsoft Tags – Microsoft MSDN Posts Ideas from the Medical Quack About Use in Healthcare!
If each lot had their own tag a cell phone could easily be used to find them. Some hospitals have RFID systems that help but they are not many so you rely on the “human” to find the lot numbers by researching the FDA and company pages to find them, and then either the same “human or another “human” to go pull the product from the shelf.
When you have the interventional surgeon doing a roto rooter type placement of a catheter in your veins you do not want a unit that could malfunction and potentially kill you.
Now after reading this please take time to vote on my poll so perhaps we can get moving in this direction. Again by using a synchronized data base with the FDA compliance on the recalls would be easier for them too. The units to be recalled were only manufactured in about a 45 day time frame, so that means there’s a lot of devices out there that are ok too, so again with a cell phone that could scan with bar codes that could be updated to show the recall on the lots affected, it sure beats relying on sales reps and notices put on the web as a sole area for information.
One more note worth the mention is that a bar code tag of what devices a patient has implanted could be stored in a PHR too, as the coding work for that is already here, just a matter of using and connecting with proper design. BD
here, just a matter of using and connecting with proper design. BD
Connecting HealthVault/Google Health and More to RazCode Encrypted Microsoft Tags – Automate Data Input
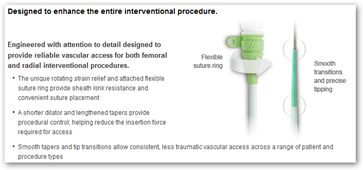
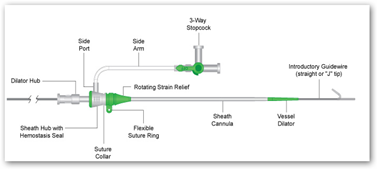
The U.S. Food and Drug Administration (FDA) has issued an urgent Class I recall of several batches of 6 French (6F) Engage Introducer devices of medical devices giant, St. Jude medical (STJ). The Class I recall is the most serious type of recall as it affects products, which can be potentially hazardous or even cause death.
The recall is based on the concern that the 6F Engage Introducer devices have the potential to cause fatal bleeding during their use. The FDA is apprehensive about the possibility of separation of the shaft from the hub or break in the hub assembly in these affected devices, which may lead to bleeding thereby, threatening life.
The Engage Introducers, which are being designed to improve interventional procedures (from vascular access to closure), were approved in the U.S. and Europe in March 2010. These devices are used to implant catheters and electrodes (electric conductors) into blood vessels during surgery and help prevent hemorrhage. According to St. Jude, the affected batches of the devices were manufactured between April 27 and June 3, 2010
St. Jude Recalls Introducer Device



0 comments :
Post a Comment