As you may or may not know I am also a webmaster who does some websites and  work with my sponsors and this one is again worth looking at as the company creates commonly used devices, in other words the nuts and bolts products used in many orthopedic procedures and with having the FDA approved products designed and made here in the US, that is a plus. In addition there’s no intellectual property rights to pay for, there’s the price difference as we as patients get a break in not having to pay for big patent expenses.
work with my sponsors and this one is again worth looking at as the company creates commonly used devices, in other words the nuts and bolts products used in many orthopedic procedures and with having the FDA approved products designed and made here in the US, that is a plus. In addition there’s no intellectual property rights to pay for, there’s the price difference as we as patients get a break in not having to pay for big patent expenses.
Disclaimer here is that I do the website and IT work here for SNNWire and this product fits right in with anything else I would blog about for savings and new developments in healthcare.
You can see the latest magazine under my sponsor section for Micro-Cap Review under my sponsor and advertising section at any time which is updated quarterly. Below is the article from the magazine which you can expand and read. Click on the “expand” button to read the entire article.
If you want more information and to watch a video interview with the CEO use this link for more information. Also there’s a back link on the Medical Quack from last year below that has additional information and a video update. Made in the USA doesn’t bad either to my ears. BD
“Generic Medical Devices” FDA Approved Doctor Preferred Orthopedic Implants Made in the USA Costing 50% Less
From the website:
“IFS Is committed to driving down the cost of healthcare by offering a comprehensive line of "Best in Class" implants at generic prices. IFS seeks to 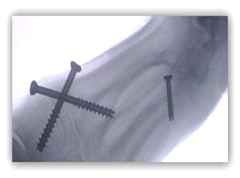 disrupt the orthopedic implant market and redefine how implants are purchased.
disrupt the orthopedic implant market and redefine how implants are purchased.
We focus on commonly used, market proven products that do not have IP protection -- 80% of what orthopedic surgeons, hospitals and surgery centers use every day.
IFS implants are designed by world-class surgeons and priced to address the declining reimbursement for surgical procedures.
Our prices are 40-60% less than our competitors.”
Internal Fixation Systems, Inc. (IFIX.OB: OTCBB) is a medical device company that specializes in products that orthopedic surgeons use every day. The company focuses on commonly used, market proven products that do not have IP protections that are designed by world-class surgeons. SNNLive met with Dr. Stephen Dresnick, CEO of Internal Fixation Systems at the OTC Markets Center in New York City.



Nice post
ReplyDelete