The procedure is not yet available in the US, but clinical trials are in place.
Her doctors told her that she had congestive heart failure and that she was too high a risk and that surgery was not an option.  "The Impella is threaded through the widest artery in the leg, known as the femoral artery. This artery gives access through the heart valve into the left ventricle -- the part of the heart responsible for pumping blood throughout the body." as quoted from the article, it keeps the blood pumping as well as the blood pressure at proper levels during a procedure. There are Established Medicare Reimbursement codes set up and the process is already used in Europe. BD
"The Impella is threaded through the widest artery in the leg, known as the femoral artery. This artery gives access through the heart valve into the left ventricle -- the part of the heart responsible for pumping blood throughout the body." as quoted from the article, it keeps the blood pumping as well as the blood pressure at proper levels during a procedure. There are Established Medicare Reimbursement codes set up and the process is already used in Europe. BD
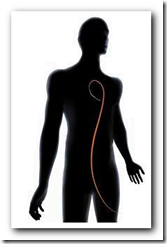 But Zeoli was being cared for at St. Elizabeth's Medical Center in Boston, which was planning to participate in a trial for a new heart device. The device is typically used during medical procedures that might require physicians to restrict blood flow in patients who already have reduced levels of it. In Zeoli's case, the device was used to maintain her blood pressure so that doctors could insert a stent to promote blood flow.
But Zeoli was being cared for at St. Elizabeth's Medical Center in Boston, which was planning to participate in a trial for a new heart device. The device is typically used during medical procedures that might require physicians to restrict blood flow in patients who already have reduced levels of it. In Zeoli's case, the device was used to maintain her blood pressure so that doctors could insert a stent to promote blood flow.
The Impella device has been in use in Europe for several years, but the pharmaceutical company ABIOMED, along with cardiologist Dr. William O'Neill of the University of Miami, is hoping to bring it to the United States to help patients in need of heart operations.



0 comments :
Post a Comment