What a mess this has been and gee if consumers, doctors, hospitals and so on  would have been able to use a smart phone to find recalls products lots, how much easier this could have been. One thing with this recall from Triad we found out they private label for tons of companies, and thus they too also had to put out recall alerts for their products, after they figured out if they were affected. This is huge snowball effect going here. You should see the lists…huge! If you shopped at Walgreens and bought over the counter medicated wipes, well check those huge number of recalled lots!! Is this not fun or what! No it is not. We still have no system. Here’s part of the original post I made back on January 7, 2011 when it started.
would have been able to use a smart phone to find recalls products lots, how much easier this could have been. One thing with this recall from Triad we found out they private label for tons of companies, and thus they too also had to put out recall alerts for their products, after they figured out if they were affected. This is huge snowball effect going here. You should see the lists…huge! If you shopped at Walgreens and bought over the counter medicated wipes, well check those huge number of recalled lots!! Is this not fun or what! No it is not. We still have no system. Here’s part of the original post I made back on January 7, 2011 when it started.
FDA Announces Recall of Alcohol Prep Pads, Swabs,  Swabsticks From Triad–FDA and Manufacturers Should Ashamed-Campaign for Bar Codes Still Stands Stronger Than Ever!
Swabsticks From Triad–FDA and Manufacturers Should Ashamed-Campaign for Bar Codes Still Stands Stronger Than Ever!
This is by far the biggest neglect of both the FDA and drug/medical device and over the counter healthcare products!! Have you ever tried to return a product to a retail drug store and be told “no” because it’s not on their authorized list!! Now some of their products are used by healthcare professionals too and I looked all over the web site and as close as I could come was to find wipes or products that remove adhesive tape so I think this is what the product recall is all about that could be contaminated. A cell phone to scan the box would soooooo simple. 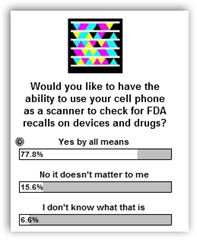
This is another good sized recall and granted the products are not expensive but for goodness sakes we have private label stuff in here now too. If in fact the products are not sanitary we need to have an easier way to find them on the shelves. Look what a mess they make for the retail stores too.
CVS and Walgreens should join my cause here as this is just simply “tech denial” and nobody wanting to do a thing about it. I have sent this to drug companies, the FDA, the DEA and several other deaf ears. This is costing the retail stores money too! Again, a good reason for them to join the cause here.
This isn’t just a J and J deal anymore and see what was on twitter about this not too long ago, everyone liked it!

I keep seeing all these notices that the FDA wants public input, but you can read the post below and see what happens when you try to contribute, its not good.
FDA Looking for Public Input on How They Communicate With the Public-In My Case They Don’t Relative to Using Bar Codes for Device, Drug and OTC Products Recalls
I guess the stuff was still shipping and being distributed, thus US Marshalls as directed by the FDA had to show up and close up shop. BD
The Food and Drug Administration said Wednesday that antiseptic products made by Triad Group and H&P Industries were seized to prevent the distribution of products that might be contaminated and pose a health risk.
At the FDA's request, U.S. Marshals have seized more than $6 million worth of products from Triad's facility in Hartland, Wis. The products included antiseptic products, cough and cold products, nasal sprays, suppositories, medicated wipes, antifungal creams, hemorrhoidal wipes, raw materials among other items. The Triad products were widely used in medical facilities around the country, and were also available for sale to the public under a variety of product names at Safeway, City Market and Walgreen's chain stores.
Triad Group and H&P Industries are owned and managed by the same parties. Triad confirmed that the seizure began on Monday and that it has halted production.
FDA seizes $6 million in products from Triad - Forbes.com



0 comments :
Post a Comment