Here we go once again and we all know the answer if you read this blog. We all know what success you have had with Microsoft Share Point and how it has increased information and efficiencies, and actually I mentioned that with my  interview with Mike Naimoli from Microsoft Life Sciences and gave Pfizer some real nice brownie points too back in 2009, so let’s think about the consumer here and offer some value here to the people who buy your products. Pfizer of course is not alone as I address any company in healthcare with recalls.
interview with Mike Naimoli from Microsoft Life Sciences and gave Pfizer some real nice brownie points too back in 2009, so let’s think about the consumer here and offer some value here to the people who buy your products. Pfizer of course is not alone as I address any company in healthcare with recalls.
A Deep Dive into Microsoft Life Sciences Today and in the Future – Interview with Michael Naimoli
The instruction are here to make a note of this lot number and then look at your product and see if you have a match. Using a cell phone to find this would be a lot more convenient and gee as a consumer I get “instant value” here with not having to do a bunch of time consuming research! I don’t this this recall is as big as the Abbott Labs one where it overloaded servers, but we never know what could be next and what drug or device, could be anyone, but the deal here is to be prepared and not be a BP of the pharma business with being prepared.
Microsoft Tag Bar Codes–Who’s Been Scanning the Medical Quack–The Bing Heat Map Tells All And Could Help Find Stolen or Expired Drugs and Devices With This Methodology
There will be more recalls in both food, drug and device products so we need a system that can work for everyone and especially the consumer as we are the ones affected. When you look at this map, and when products were scanned, guess what, it could give Abbott even more intelligence in knowing where the product was due to “spots” on the map so they in turn could be more pro-active here too with finding the product quicker! What technology that is available today for implementation can do and save lives too.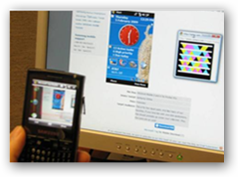 Click on the Bar Code Campaign image to read more.
Click on the Bar Code Campaign image to read more.
Here’s a hint from a Mom who recently had to check products from another company, she wanted the convenience and value. The technology is here today and needs to be designed and requires collaboration of the FDA, DEA and other government entities and a pilot program would sure be nice to give this a try. Also I have a poll going here and consumers seem to like the idea too, look at the votes! BD

The company said it is taking this precautionary step after finding a potential for a leak of the components contained in the wrap, which could cause skin injury such as irritation or burn. The issue is limited to the recalled lot. No other ThermaCare products are impacted. Pfizer has notified the U.S. Food and Drug Administration.
The lot number of the product involved is:
Lot #
Exp. Date
Product Name
E06831
8/2012
ThermaCare HeatWraps Menstrual
This lot number is listed on both the outer carton and the foil pouch. The lot number may be followed by either an S or an N. For more information on the recall and where to find this information on the package please visit www.thermacare.com or call 1-800-323-3383, Monday through Friday, 9am to 5pm EDT.
Pfizer Consumer Healthcare is removing the product in question from store shelves and asking consumers who have purchased and are still in possession of the affected product to record the lot number, throw the product away in its entirety without opening the foil pouch, and call 1-800-323-3383 for replacement.



0 comments :
Post a Comment