The reports on the device state that it contributes to both weight loss and normalization of blood glucose levels. In the trials the device was in place for 12 weeks, and then removed. The procedures takes around 30 minutes or so and is done endoscopically through the mouth. The report stated that participants lost triple  the amount of weight normally lost with diet programs.
the amount of weight normally lost with diet programs.
The patients with diabetes 2 experienced decrease in blood glucose levels and a reduction in diabetic medication. This appears to be some exciting news on the war against obesity with a temporary device to help individuals lose weight and could be a viable alternative to the more invasive bypass surgery.
The product is in clinical trials at present time and represents one more example of the move toward devices versus drugs in healthcare. The fact that glucose levels drop and a reduced need for diabetic medication also is certainly very promising. When seriously obese patients need an MRI for example, sometimes the procedure can’t be done due to patient size and weight. BD
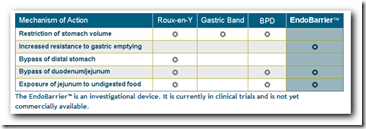
“Because it is designed to mimic the effects of gastric bypass surgery, the same metabolic control of diabetes documented after surgery may be realized with the EndoBarrier™. Eighty-four percent of patients immediately resolve diabetes with gastric bypass surgery, a phenomenon found to occur as a primary, specific, and direct effect of this surgery, not secondary to the weight loss. “
GI Dynamics, a medical device company pioneering the development of new approaches to treat obesity and type 2 diabetes, announced today a paper appearing in the advance online publication of the journal Obesity, which demonstrates that implantation of an innovative endoluminal sleeve mimics key, beneficial effects of Roux-en-Y gastric bypass (RYGB) surgery, inducing substantial weight loss and improved metabolic function in rats with diet-induced obesity. The dramatic effects of the removable device on blood glucose levels in this animal model suggest an important role for the duodenum (the first part of the small intestine) and jejunum (the central part of the small intestine) in the regulation of this metabolic function. GI Dynamics is developing an endoluminal sleeve, known as the EndoBarrier" Gastrointestinal Liner, and is currently in late-stage clinical trials.



0 comments :
Post a Comment