I first wrote about the implantable device back in 2011 and today it has the European CE approval and working closer with the US for FDA approval. The funding will pay for the rest of the clinical trial process. The device uses vibrating or other alerts to get the attention of the patient and it stores ECG traces as well. From the website you can read more about the “ALERTS” study…
“The ALERTS study design provides a clinical environment to test the efficacy of a device that may significantly reduce time to 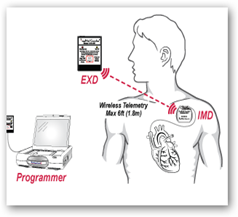 treatment and thus may decrease heart muscle damage and improve survival rates related to major cardiac events. After completing a Phase One study in the US, the AngelMed Guardian System is presently being tested in the ALERTS pivotal study. The ALERTS study is a prospective, randomized multicenter study with FDA approval for 100 centers and 1020 subjects with a high-risk of having a heart attack. All subjects have data collection and analysis enabled from the onset, however, subjects are randomized 1:1 for patient alerting ON or OFF for the first 6 months. After the initial 6 month period, patient alerting is enabled for the control group. All subjects will be followed until study closure.”
treatment and thus may decrease heart muscle damage and improve survival rates related to major cardiac events. After completing a Phase One study in the US, the AngelMed Guardian System is presently being tested in the ALERTS pivotal study. The ALERTS study is a prospective, randomized multicenter study with FDA approval for 100 centers and 1020 subjects with a high-risk of having a heart attack. All subjects have data collection and analysis enabled from the onset, however, subjects are randomized 1:1 for patient alerting ON or OFF for the first 6 months. After the initial 6 month period, patient alerting is enabled for the control group. All subjects will be followed until study closure.”
AngelMed Guardian System-Implanted Device to Alert Patients They Are Either Having or Are Close to a Heart Attack-Clinical Trials
Angel Medical Systems, an innovator in today's cardiac medical device arena, recently celebrated the implant of the 600th patient with its AngelMed Guardian system. With the help of its manufacturing partner, Flextronics, Angel Medical Systems also achieved this milestone more than two months ahead of the company's estimate.
AngelMed Guardian is designed to detect and alert  patients of acute episodes of cardiac ischemia related to the progression of coronary artery disease and thrombotic coronary occlusion caused by vulnerable plaque ruptures. Some of the external monitoring devices that are part of the Guardian's alerting system were built by Flextronics in Valencia, CA.
patients of acute episodes of cardiac ischemia related to the progression of coronary artery disease and thrombotic coronary occlusion caused by vulnerable plaque ruptures. Some of the external monitoring devices that are part of the Guardian's alerting system were built by Flextronics in Valencia, CA.
http://www.onlinetmd.com/angel-medical-systems-implants-cardiac-device-12312.aspx



0 comments :
Post a Comment