There are many stents and stent stories out there, but this appears to be the first of it’s kind for the US, as the product has been approved for use in other countries for over a year now, such as India and Israel. As noted, the product received an award for being a revolutionary stent with the  tissue covering.
tissue covering.
A while back I had the opportunity to discuss stents with Dr. Muhs, Assistant Professor of Vascular Surgery and Radiology Co–Director of Endovascular Surgery from Yale University. He had practiced overseas and stated we are a bit behind the rest of the world with approving new developments and products in this area, and it was really nice to arrive at Yale with having prior experience in other countries with products already being utilized.
One of the target areas for the new stents is for treating aneurysms. Dr. Muhs is also an interventional radiologist whereby minimally invasive procedures are mapped via images, to plan the course of action and navigation to target the affected area, in other words with software imaging the surgical procedure is fully planned and charted out before beginning a procedure. If you need this type of surgery, you are going to want the interventional radiologist involved too. I also had an interview a while back with Cook Medical and their new efforts to support an entire business unit devoted to interventional radiology.
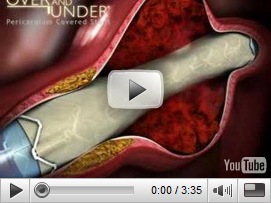
In order for hospitals to have many types of stents on hand, they are not charged until the stent is actually used, and with the cost, and RFID tracking system for the tiny items is definitely in order. The video below is probably one of the best animations I have seen showing how the stents are placed. Also the website has additional information on the process. BD
ITGI Medical Ltd
and its subsidiary Design & Performance (DPC) are a bio-medical engineering group specializing in the research and development, design, manufacture and distribution of heterologous tissue covered stents and products for vascular applications.ITGI Medical Ltd. (TASE:ITGI) has obtained US Food and Drug Administration (FDA) approval for two cardiovascular stents as humanitarian use devices (HUD). The Or Akiva-based company's share jumped 320% to NIS 0.21 on the news.
ITGI obtained approval for its pericardium covered Over and Under Stent and its Aneugraft Stent. Both stents are already marketed in Israel, Europe, and Latin America. In 2008, the company won a Frost and Sullivan award as a "revolutionary stent company".
Stent co ITGI Medical soars on FDA nod
Related Reading:



0 comments :
Post a Comment