Clinical Trials are just finishing up here in the US and the company is hoping to receive FDA approval perhaps in the 3rd quarter of this year. It heats the airways and is a minimally invasive  procedure with going through the mouth or nose.
procedure with going through the mouth or nose.
There is no incision and if you have watched some of the cardio catheter videos, this may remind you of how a catheter or device travels, guided by imaging. This could be good news for those with asthma who are not getting relief from medications or have side effects. 3 treatments are estimated to treat an entire lung. After treatment, patients have more symptom-free days, so this is a long term type of treatment. BD
About Bronchial Thermoplasty (website)
- Bronchial Thermoplasty is a new investigational outpatient procedure that is performed in the hospital bronchoscopy suite.
- Bronchial Thermoplasty is currently performed only in conjunction with the AIR2 Trial.
- The objective of the procedure is to reduce the airway smooth muscle in all airways reachable by the bronchoscope in hopes of reducing the airways ability to constrict.
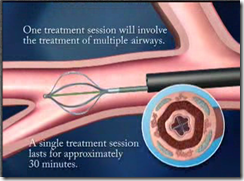
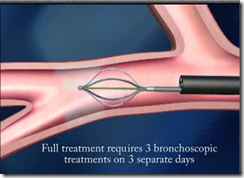
- A Bronchial Thermoplasty treatment session is expected to take less than an hour to complete; it is expected that three treatment sessions will be required to treat the entire lung system.
- Bronchial Thermoplasty is an outpatient procedure. No general anesthesia is used, no incision is necessary, and there is no need to stay overnight.
CHICAGO (Reuters) – An experimental asthma treatment that uses heat to reduce airway constriction provided some relief from severe asthma that is poorly controlled with medications, U.S. researchers said on Monday.
They said the Alair device, made by privately held Asthmatx Inc of Sunnyvale, California, cut the rates of extreme asthma attacks by 32 percent and reduced trips to the emergency room by 84 percent in patients with severe asthma.
Patients missed fewer days of work or school because of asthma symptoms and had more symptom-free days compared with people who received a placebo, according to results of the late-stage clinical trial, which was presented at a meeting of the American Thoracic Society in San Diego.
Asthmatx Inc is seeking U.S. Food and Drug Administration approval for the device, Castro said, and a decision is expected this fall. The treatment has been approved in Europe.



0 comments :
Post a Comment