The therapy is not cheap and there’s also the seeding process involved. The seeds draw the radiation rays to the cancer cells, thus leaving the healthy surrounding cells untouched. This technology goes on step further and is able to adjust the radiation based on the location of the prostate gland, in other words when it moves, it can adjust the area of focus immediately with the technician turning off the machine or making an  adjustment on the direction of the rays with the table, thus again, less radiation exposure to areas where there is healthy tissue. The procedure is used at the Cleveland Clinic and several other hospitals. By exposing healthy tissue nearby to the radiation, it could make the patient impotent or incontinent.
adjustment on the direction of the rays with the table, thus again, less radiation exposure to areas where there is healthy tissue. The procedure is used at the Cleveland Clinic and several other hospitals. By exposing healthy tissue nearby to the radiation, it could make the patient impotent or incontinent.
A similar procedure is done for breast cancer, called the Mammosite, that also involves “seeding”.
The economy has taken it’s toll on the company with a few recent lay offs. With having insurance or Medicare covering the procedures and making it available to patients, that certainly can’t hurt the health of the consumer or the company. This is always one big concern with cancer treatments, whether it is paid for and how available the therapy is for the patient.
Calypso Medical Technologies has an expensive new technology for targeting radiation beams to cancerous prostate glands, so it shouldn’t be any surprise when insurers balk before they pay up. But Calypso got some good news this week as it persuaded an important customer, the regional Medicare unit that serves a lot of elderly men with prostate cancer in Florida, to set up a standard reimbursement practice.
The Seattle-based medical device company currently has established Medicare reimbursement practice codes in eight of the 15 regional units around the country, and has gotten reports from medical centers in 12 states that they have actually gotten paid for their daily use by Medicare, the company said. Calypso also has established reimbursement from more than 13 private sector insurers, including many of the big names, like UnitedHealthcare (NYSE: UNH), Humana (NYSE: HUM), Cigna (NYSE: CI), and several regional Blue Cross/Blue Shield units.
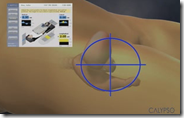
This whole step-by-step process of reimbursement, and the time it takes, has made it clear that simply winning FDA approval may no longer be the automatic ticket to the promised land for upstart medical device and biotech companies. Calypso’s technology, which it markets as “GPS for the Body” was first approved by the FDA in August 2006. The system uses transponders implanted in the prostate, which send a signal to a base station that tells a technician precisely where the prostate is in the body in real-time.
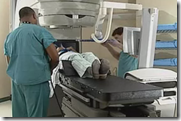
By tracking the prostate this way, Calypso’s method is supposed see whether buildup of urine in the bladder or gas in the rectum has caused the patient to fall out of careful alignment with the radiation beams. When the beams are off track, they can zap healthy tissue nearby that makes men impotent or incontinent—which most guys I know would rather avoid.

Calypso Gets Florida Medicare Unit to Pay for “GPS for the Body” for Prostate Cancer
Related Reading:







0 comments :
Post a Comment