If you have been a reader here long enough at the Quack then you have probably read one of my articles about the use of bar codes on drugs and medical devices. We have somewhat of a step in this direction, but lots of lack luster still missing. You can read the short paragraph below and see it is aimed at “marketing” and little mention of safety with over the counter drugs. The product they came up with here is a very small version of what could be done.
“Barenburg also sees potential for its watermarking technologies in allowing companies to engage with their customers, through use of the code to link to promotions, coupons and other materials, for example for over-the-counter medicines.”
Microsoft Tags – Microsoft MSDN Posts Ideas from the Medical Quack About Use in Healthcare!
For one, there’s no Heat Map to find the counterfeit drugs nor are we even mentioning recalls so again we have a marketing focus and the consumer once 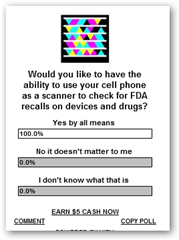 more is getting a little information from what’s left over, enough to keep consumers happy in the long run, don’t think so. This is for over the counter only and there’s no mention of including the FDA for a synchronized data base for compliance measuring, something I feel is important but so far ignored by the FDA and DEA.
more is getting a little information from what’s left over, enough to keep consumers happy in the long run, don’t think so. This is for over the counter only and there’s no mention of including the FDA for a synchronized data base for compliance measuring, something I feel is important but so far ignored by the FDA and DEA.
If you have a chance enter your vote before you leave this page and let me know if you would value being able to use your cell phone as a scanner to find recalled products, drugs, devices etc. right on the shelves.
Microsoft Tags in Healthcare – Comments and Demonstration at “Microsoft Connected Conference” with Surface
Now with this data base they are creating, why not put a Tag on each page and have the device and drug companies synchronize their updates with the FDA? This also gives the FDA an answer to monitoring compliance with checking to see if the “tags” have been updated, what a novel idea use some technology here, right? This is not much different than faxes were in the early days as we had people skeptical about those too and now look where we are.

In addition we have bar codes that do only “one thing” so we come back around to that wonderful word, collaboration. Hospital tech folks like the Tags as they can work with RFID systems too. In addition, Microsoft Tags can be stored in a personal health record too and have the potential to authenticate for e-prescribing and thus I think again we are missing a huge part of the picture here.
RAZCODE (Microsoft Tags) Using Smart Phones to authenticate MDs When e-Prescribing Controlled Substances
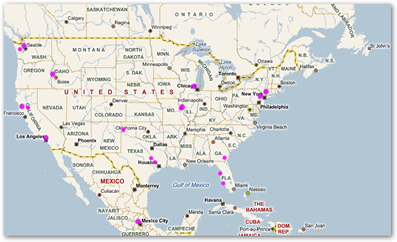
They have the GPS tied in here but again where’s the map? There’s also no inclusion of any type of medical device mentioned and those are recalled too by the numbers so hospitals are left out here on verifying what’s on the shelf and people die getting implanted with devices that have been recalled and miss getting pulled from inventory. Here’s an example of a heat map that shows who has scanned my bar codes as listed on the blog.
Look at the mess created by Abbott with the glucose testing strips and there have been glucose devices recalled too, again the job is only half cooked here with not getting devices in the game.
Abbott Diabetes Care Recalls Tons of Glucose Test Strips–The US Has the FDA Recall Blues-Solution Has Been Touted Here for a Year-It’s Time for a Fix–Readers Have Voted
The article also mentioned Best Buy and the use of Canon with bar codes. I have gone into the stores and checked it out, and the QR codes didn’t work but the Microsoft Tags with Canon did work, and very well.
Smartphone Scanning Coming to Best Buy and Staples on Canon Products–FDA Recalls In This Same Fashion Would Be Nice With Microsoft Tags As We Have No System Right Now
Again, this is a nice gesture of getting started by a long way from where it could be with an appropriate business model that could cover so much more and add a little emphasis on safety rather than the full on marketing we get today. This is what consumers want, safety. In the meantime, the FDA goes about their antiquated methodologies, which is better than nothing to get the word out on recalls.
Collaboration is just buzz word that everyone uses for their marketing today but nobody does squat to pursue safety technology. I think this is post # 62 that I have made in a little over a year’s time. This again does nothing for devices like this stent recall.
Stent Catheter Recall-NanoCross Dilation Catheter Could Break During Use–Need Bar Code Technology to “Scan that Catheter”!
In summary here this is an attempt but lacks the full on use that could be realized and doesn’t do enough for the overall picture of solving many problems with one technology. BD
A pharmaceutical manufacturer has started a product serialization pilot using a coding system developed by US firm Mobile Data Systems, with each code protected using the firm's digital watermarking technology.
MDS sees great potential for its technology in the pharmaceutical sector as a means to guard against counterfeiting and diversion, implement track-and-trace to help meet upcoming pedigree requirements in the USA, and assisting recalls and returns.
The pharmaceutical pilot will include 30,000 to 40,000 individual packs of medicine supplied by the 'major' pharmaceutical manufacturer, according to MDS director of business development Ron Barenburg.
Codes will be added to the artwork of the pack, label and package insert and the test products will be shipped through the pharmaceutical manufacturers' usual supply chain with scanning and authentication of the codes at various points, including distributors, wholesalers and pharmacies using smartphones provided by MDS.Using a smartphone or camera phone with the appropriate software, players downstream in the supply chain can scan for the FadeMark, whereupon a server records the phone information, GPS location, time and agent information. If the FadeMark is detected and is valid, confirmation is sent back to the agent, otherwise the agent receives an alert that all is not well.
SecuringPharma - Drugmaker gears up for pilot of MDS' coding platform




0 comments :
Post a Comment