This is very nice, no more having the “hearing aide” visible and it is implemented behind the ear. The clinical trial was conducted with 61 individuals, which by clinical trials numbers is small, but the results seem to speak for themselves. You can visit the website and listen to several videos from people who have the implant and how it works for them. There’s even Bill, a pharmaceutical sales representative who has one. Bill says it was difficult to make sure he heard the orders properly so we don’t want areas of such to be missed when making your sales calls.
He talked about being able to go into the water now without having to worry about removing his hearing aide. Bill the pharma rep said he thinks his hearing problems put him out of touch with being able to advance within his company. He should be seeing higher sales now I guess:)
The battery is slated to run for 9 years so at some point in time there’s a visit back to the doctor for a battery change, or by that time perhaps a new type of advanced device may be available too, and I think that last statement is the reality of what we are seeing in device research and development today. The device is for those who are impacted severely or moderately with hearing loss. BD
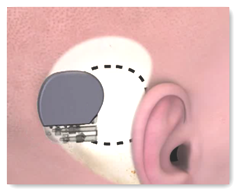
The advanced implanted hearing system is the revealed to be the first completely implanted system which has grabbed an approval by the U. S. Food and Drug Administration for adults suffering from moderate-to-severe sensorineural hearing loss, a permanent lack of hearing.
The system make use of sensors, processors, and drivers to convert vibrations in the eardrum and middle ear bones into electrical signals that are finally amplified and filtered to compensate for the losses in the sounds and speech signals.
The FDA approval is initiated based on a multicenter clinical trial involving 61 patients comparing Esteem to pre-implant hearing aids, revealing that nearly 56% of them scored better with the new device, with a net 93% scoring beating, in line with the initial scores on the test.
FDA Approves First Totally Implanted Hearing Aid | TopNews United States




I'm one of the patients in the clinical trial, and I can honestly say it has changed my life. As someone who was never comfortable wearing hearing aids, for several reasons, the Envoy Esteem seemed like the perfect solution to my hearing problems.
ReplyDeleteSince I've been implanted, I've become much more socially outgoing, and I no longer struggle to communicate with others. It has truly been a blessing, and I hope my daughter can one day benefit from this amazing technology!
I have a blog about my experience if anyone would like to learn more: http://alreadygone78.livejournal.com/
=)
Great post ... really helpfull
ReplyDeleteThank you
Regards,
Jack from Hearing Aid Reviews
Great post. I really like it so much.
ReplyDeleteThis really looks good. May I know if this has any negative impact to the person who had the implant?
ReplyDeleteI love new technology with hearing aids. My son has hearing aids and is very young, but the advances in technology are so amazing! I wonder what will be available in another 10 years or so? The fact that the Esteem is waterproof is a great advance.. now if only insurance would pay for it!
ReplyDeletehttp://hubpages.com/hub/The-Esteem-Hearing-Implant-An-Invisible-Hearing-Aid-Alternative
Is it really comfortable to have one? I agree the advancement in the technology of hearing aids are getting greater and greater.
ReplyDeleteThis is all well and good, but what do they cost? This is where I have a big problem. The government allows companies to develop these devices and charge whatever they think they can get away with. As the technology improves, unlike electronics, the costs go up, not down. And as one person commented, insurance does not cover them. Both hearing devices and glasses are an essential part of a person's total health and should be subsidized by the government to make them affordable. Just like the drug companies, who also make huge profits at the expense of ordinary citizens, the hearing device and glasses industries do the same. Instead of subsidizing big oil companies, why doesn't the government subsidize the health industry, including drug companies, hearing device companies, and glasses manufacturers?
ReplyDeleteI will continue to not wear a hearing device as long as I possibly can, or until I get too old to care any more.
Generally, the cost of the device itself, the implant procedure (including the cost of the implanting surgeon and his or her staff, the surgical facility, and the administration of general anesthesia), and the necessary return visits for turning on and programming the device to fit your unique needs will be approximately $30,000.
ReplyDeleteDrug companies make such "huge profits" that as an investor I much prefer investing in CocaCola. It is a much safer investment. The millions coke invests in product research always results in a return, whereas the millions invested in researching drugs only result in a return for 1 out of 5 drugs that actually work as desired and get to market. I can't wait to hear your new complaints about the insane profits of CocaCola.
ReplyDeleteMaybe I can get the government to subsidize my Coke. Along with my gas and oil - I have noticed that when I can't drive anywhere my quality of life is GREATLY diminished!
"If" hearing is a priority in your life then save for the implant. "If" seeing is a priority in your life then put some money aside for glasses or contacts. "If" a flat screen TV or a sports car or a trip to the Bahamas are priorities then work hard, save and go for it! Saying the government should pick up the tab is the the same as asking your friends and neighbors to pick up the tab...really?
ReplyDeleteI have severe hearing loss and if I think the Esteem will help I will find a way to get it done.
I understand both sides of this argument, but do you realize that many group health insurance plans actually pay for VIAGRA? That is nuts! If you want the little blue pill, go buy it! It is NOT a must have medication! I would think hearing and seeing would be considered MUST HAVES way before Viagra! People who can't hear or see very well not only have negative impacts on their lives but on all of our lives. Consider the fact that you may be hit by someone who can't hear you honk your horn!
ReplyDeleteHopefully the cost of the Esteem procedure will come down to about half of today's $30,000.
ReplyDeleteMy wife has severe hearing loss and the several hearing aids we have purchased over the years costing about $4,000 each have been of very little benefit. I could afford $15,000 for the Esteem procedure, however I doubt if we could be given a payment plan for the balance at our age.
There is one thing not addressed here... The cost is around 30K for the surgery, BUT what about post surgeries to replace the battery every 4.5 to 9 years? What is the cost of those? And more importantly, how is the effect of repeated surgeries on the ear over time? What about scar tissue? All of these things should be a consideration BEFORE the implantation. So I would appear that this is not an end all solution for hearing loss. I have worn hearing aids for about 10 years now, and I would NEVER go through this surgery for the reasons stated above. I would rather wait until there is a one surgery option that is safe.
ReplyDeleteVery curious about this. I had spinal meningitis when I was almost 5 years old and have been profoundly deaf since then. I never used hearing aids cuz they didnt work. I speak well and lip read well through speech therapy in my younger years. I am now 44 and have known about the cochlear implant for years, but have been skeptical about them. If they had trials for some of their newer stuff, where I dont have to worry about costs for new surgery or batteries, I'm all for it. I like the fact that I could use it wherever I am, including the shower or pool. This is what I have been waiting for for a long time now. Lets hope the price comes down and/or their batteries can last much longer or they can give us a guarantee of 10 years or the next battery is free, including surgery etc.. like we get for our cars..
ReplyDeleteI just love it when people think they are owed something from the government. We wonder why the country is falling off the edge.. I am going to go thru the procedure middle of next year. No insurance. I am finding a way to obtain the funds on my on.. It's my hearing loss. I have worn hearing aids for 21 years and now they are no longer effective. I am nearly deaf at 62. I want to continue working and teaching. This will hopefully be the answer. But, the lack of insurance or government subsidies won't keep me from trying to get my hearing back. If it's important enough to anyone, the money shouldn't be the roadblock.
ReplyDeleteI am 48 and have had a sudden hearing loss to my left ear and use a hearing aid in my right ear. Without my hearing aid, i am deaf. I am saving for the implant! I am excited that there is such a solution out there. After i pay for the 1st one, i am definitely gonna save for my right ear. The thought of hearing normal again and swimming without being deaf is just so exciting to me! I can't wait! I want to hear again so badly! I am missing out on so much and i feel like it's effecting all my personal relationships along with my family relationships. Everyone's irritated with me being deaf. Thanks Esteem!
ReplyDeleteTo: Eric M. emendez777@yahoo.com....Re: Cochlear implants....NOTE: If you're a liberal, please dont freak at the name "Rush Limbaugh" (People frequently do...) This has nothing to do w/politics.... Eric: Look into the cochlear implant; talk to a doctor. Rush Limbaugh went totally deaf some years ago (in only months), from an auto-immune disease that attacked his hearing. Once he'd had his surgery, he told his radio audience abt everything: how he went deaf, what it was like, search for a diagnosis, & his cochlear implant: every step of the process, the surgery, adjustments, how he fiddled w/it to get it right, the whole thing. Short version: the implant enabled him to continue his radio broadcasts. He can carry on normal conversations & hear his callers. Drawbacks he noted: Music doesn't sound the same - he can't hear the underlying layers. TV is hard to totally understand, so he uses close-captioning to help him understand what's being said. Noisy parties are difficult. But w/it, he can live a normal life. Cochlear implants are for the totally deaf & connect w/the Inner Ear; Esteem is for if you still have some hearing & connects with the Middle Ear (your eardrum & such still work).
ReplyDeleteHi Everyone,
ReplyDeleteI'm an audiologist practicing in CA. I work for/with a very good ENT who just was down at the House Clinic in So. CA re: this device. Honestly, this technology looks amazing. However, as with anything new, there are sure to be bumps in the road. Or for that matter, people with hearing loss where this device will not be effective or appropriate. For instance, I saw a post earlier from someone who had meningitis and for them it likely wouldn't be any more effective than hearing aids since the cochlea is not functioning (possibly/possibly ossified). In fact, for anyone who has siginificant damage to their cochlea (specifically the hair cells on the basilar membrane), the problem is the individuals ear and not the delivery mode (Esteem vs traditional hearing aids). Plus, it is significant surgery in a very tight and delicate area with all of the incumbent risks that entails.
But there is no question that this technology is pretty amazing. The price factor obviously is a huge downside for all but a few since it would undoubtedly be an elective surgery. But, as another poster said, if you want it bad enough it's a question of priorities. I don't think post implant surgeries will be needed in all but a few cases and replacing the battery likely would an out-pt procedure.
Bottom line though, this is just a really (really!) cool hearing aid. Without a doubt, it addresses most/all of the issues I end up dealing with in my practice: cosmetics, ease of use, care, occlusion, feedback, compliance, etc. If they can bring the total cost down to about $15k, this is definitely a huge step.
I lost hearing in my left ear due to overnight hearing loss about 4 years ago. Initially I did not have any hearing at all, but after a lot of prednisone, I did regain some hearing. I can hear loud sounds but have great difficulty with word recognition, sound placement etc. I am familiar with Esteem, and would defintely find a way to pay for it if it would work for me???
ReplyDeleteWill this be covered under "ObamaCare"?
ReplyDeleteI would like to hear from people that have or are using the Lyrics hearing system. I am currently using the Lyrics and am considering changing to the Esteem. Any advice or comments?
ReplyDeleteI had the esteem done 7 months ago and it has not worked so far. I am 37 years old and was told that I was the perfect canidate. I am still hopeful but now I am beginning to lose hope for my appointments have been put off again. I really pray that this isn't the end for me. I am a single mom of two young children and we are all dying for this to work.
ReplyDeleteI had the implant Feb. 2012 after four trips back to Chicago Dr. Marzo removed it saying it wouldn't work for me. Now my hearing in that ear is not as good even with a new hearing aid .
DeleteReally? I would hope you would post all comments so we can have a true picture of what could happen.
ReplyDeleteI would just like to point out that not everyone has the money to just save 30,000 LOL. So I find it funny that people are saying if your priorities are straight then you will find a way to get the cash. As mentioned this procedure is not paid for by insurance companies either. I would also like to point out that there are huge scholarships and grants for other medical problems and prosthetic devices. I guess I am crazy though huh.....A person can do without hearing but cannot function without a leg or an arm. Not everyone can use hearing aids by the way. Did anyone ever consider healthcare professionals ? WOW imagine the trouble a nurse or a doctor would have wearing hearing aids with a stethoscope. Well I speak from personal dealings......I am unable to wear aids at work with my scopes and I cannot afford this procedure. I can see where the above mentioned person would think there should be assistance provided in some way. I guess all the money going to welfare recipients that have been on it 10 years is a must right ? I am a hard working single mother that could use some help. I pay my taxes and though I do not want handouts it would be nice to see some help for disabilities instead of handouts being given to people that do not need it. I believe the true issue is the insurance companies here along with the drug comapanies. maybe the government could step in there instead of constantly stepping onto foreign soil to always lend a helping hand.
ReplyDeleteThe Anonymous dated Jan 5, 2012. What damage did you have to your hearing loss. nerve ending hair cells or nerve audio damage? My husband is seriously considering this procedure in the next month. Not sure now after seeing your post. Would love to talk with you on the phone. Thank you and God Bless
ReplyDeleteAgain for the post dated Jan. 5, 2012. Did they tell you what might be wrong? Are the Esteem people trying to work with you? I am having the surgery soon, and would like to know more about your experience.
ReplyDeleteSorry I did not see your post earlier. I would love to talk to you. My name is Jennifer and you can reach me on my cell phone at 440-413-0719. I was told that I was the perfect canidate...so not sure what went wrong. Yes, they are working with me but I think it is going to be that it did not work. They really haven't said yet. However, they said this was the best it was going to get. I am still trying to wrap my head around this and trying to figure out what to do next. Where are you having this surgery? Which engineer will you be working with? I am available to talk anytime after 3:45 pm. I hope and pray for you and want this to work for you. after reading all these posts about the successes, I was extremely upset that I will not be able to share the joy as well. My biggest reason for doing this was for my children. Let me know if I can be of any help in answering your questions.
ReplyDeleteBe very cautious people. The Esteem folks are not very cooperative.I sent my audiogram and they told me I was a good candidate. I had to send money to talk to the surgeon, so I did. I wanted to go forward and was ready to raid my 401K to pay for the surgery. The surgeon called, but got very frustrated that I couldn't comprehend on the phone, even tho he knew in advance it would be problematic. He offered to e-mail me. I waited a month, but he never contacted me. I even flew to Houston and met with PR folks and a surgeon-in-training. Suddenly I was persona non gratia. No one would speak to me, no one would return my e-mails. After some angry and bewildered e-mails, finally, someone in so-called customer service said I needed to get a cochlear implant evaluation and send the results. I did as requested. They informed me I was no longer a candidate and returned my deposit. They refuse to explain why all contact ceased; they refuse to explain why I'm no longer a candidate. I'm left to wonder if I truly am not a suitable candidate or if I offended someone and they simply don't want to be bothered with a "problem" patient. Seems like I'm entitled to an explanation . . . So, be wary folks.
ReplyDeleteI now have both ears with the implant. It has changed my like in many ways. Check with your insurance to see if you can get a suppliment policy that covers experimental operations such as this. My supplimental policy paid 100% of both operations. Those of you who were with AT&T should have this care plus coverage. It is well worth it. I saved for the surgery until my insurance told me it was covered due to the supplimental policy I had. What is quality of life (hearing/seeing) worth to you? The cost of the batteries in 4.5-9 years is also covered. ($5-$6K)
ReplyDeleteJoe in Houston, could you please elaborate a bit on your results? How bad was your hearing prior to implantation?
ReplyDeleteAlso, Joe in Houston, what is the name of the insurance company that sold you the supplimental policy?
ReplyDeleteHi everyone
ReplyDeleteI had the Esteem implant last April. I started a blog about it because I was unable to find anything except the glorious testimonials on Envoy Medical web site. You can find my blog at kayesteemhearingimplant.blogspot.com. I invite anyone to post comments on that blog to give others their experience and to ask questions. We need to hear about both good and bad experiences. I will say this, that the Envoy people and my Esteem doctor are treating me very well. You may email me at kay.green169@gmail.com
I am also interested in Joe's hearing loss level before the implant.
ReplyDeleteI so appreciate the time people are taking to share their experience and concerns with the Esteem middle ear implant. Please post more and with specifics. I have moderate severity SNHL and have been investigating the Esteem extensively (talked with Envoy reps, emailed receipients NOT on the website, and looked at excellent blogs like Kays). Not all people have had a good experience; specifically, if you have severe SNHL the Esteem people are giving the go ahead and they KNOW the results aren't so rosy with severe level of SNHL. I may very well get bilateral Esteem implants, but it breaks my heart that people are forking over BIG money and getting suboptimal results. As an audi from California said in a post above, it is in fact an implanted "hearing aid" with a slightly different mechanism than traditional nonimplanted aids, so improvement on average is about 5-10 decibels with some improvement in speech recognition (see the study results from the Esteem trials). Living with hearing loss is not easy (read: emotionally painful).
ReplyDeleteHad my implant surger on Jan 10, and my device turned on yesterday. The implant is so much better than hearing aids that I have worn 30 years. The Envoy people are fabulous and go to great lengths to answer questions and help. Quality care! My doctor was from California, I live in Texas.
ReplyDeleteI had the implant surgery last week so I am still recovering. So far so good. Barb, I am assuming you had your surgery in the Woodlands? If so, I did too. I was highly impressed with the facility and the staff. They were amazing and very helpful.
ReplyDeleteWould love to hear more feedback from Joe and Jennnifer and any other implantees. Thanks
ReplyDeletedoes anyone else find it odd that many of the videos on the envoy/esteem website do not have closed caption?
ReplyDeletehello everyone
ReplyDeletedoes anyone here have Alport's syndrome.My hearing loss is due to aport's and they too told me i was a good candidate for the esteem.However i have ringing in the ears as well and not sure if this would work out well before forking out that kind of dough.i live in canada.
I would like to see some other people who had the esteem and worked for them (see their audigram(
I have sensorineural hearing loss and I have been continually in debt for hearing aids over the past 50 years of my life. Each time the hearing aid people state how much "better" the latest model is....largely, it has become like snake oil, always a disappointment. It is sad that you are punished with having the disability, and then punished again for the price of technology. I am a single mom, and I would need bilateral implants. I just don't have $60,000 in my hip pocket!! I would love to find a way to make these implants possible, but a loan just isn't realistic.
ReplyDeleteI recently had the implant done at the San Jose clinic. Post surgery has been pain free with minor discomfort. Being deaf in one ear has affected me more than I thought, but hopefully the short term problem will end when they turn it on. My hearing loss is sensorineural in the high frequency range. I also have tinnitus which will be bothersome during this two month period.
ReplyDeleteMy wife is getting ready to see if she is a candidate with Dr. berry hill in Oklahoma City. Has anyone had this done there? Missouri mom
ReplyDeleteI had Dr. Berryhill but my surgery was in Houston. I can't speak for OKC but as for Dr. Berryhill, he was wonderful. He put me and my parents at ease. He was a very professional doctor and passionate about the Esteem implant and what it does for people with hearing loss. I highly recommend him.
ReplyDeleteDid it work as well as they claimed?
ReplyDeleteSorry, I haven't checked this in months. I had my device activated in May. Overall, I am very pleased. It DOES take some adjustment and patience. After not hearing certain things for over 35 years, it takes a while to get used to certain sounds and how loud some noises can be. I love the fact that I can swim and shower and actually hear and not have to worry about wearing a hearing aid, etc. I am debating on whether to get my left ear done also. I still wear a left aid sometimes. I have people all the time as me if I would go back to a hearing aid and I would absolutely not.
ReplyDeleteWell, I see I'm not the only one suffering from sensorineural hearing loss. My hearing loss is congenial and is at an present loss at about 70 decimals in both ears. After being made to jump through the hoops from Esteem with paper work, raiding my 401k, doctor visits, faxing all the hearing test results and pre-op work, I'm getting implanted August 22. Oh, the cost 30 grand? Try more like 37 grand when all is said and done with some financing. Over the past two and a half years I've spent 15 grand on hearing aids because a certain hearing despenser wasn't selling a strong enough hearing aid in the first time. So I'm hoping this might do the trick. How do I afford it? Well I make O.K money and just live like a scumbag living well below my means.But don't expect Uncle Sam's help. Obamacare will turn a {hahaha} deaf on you. Also the hearing care industry doesn't have the muscle to grease the hands and wheels in the Washinton beltway to make insurance companies cough up the dough either.Does anybody else feel like they're not being heard either?
ReplyDeleteI m in Chicago area. I was told back in April that i was a candidate for esteem implants. Dr. Marzo told me they are waiting to get approval to do esteem surgeries in Loyola hospital in Maywood, IL instead of Oakbrook surgical center in Oakbrook, IL. I waited 4 months and all of a sudden Dr. Marzo decided not to implant esteem because I have profound high frequency loss (I did 52% in speech test though, requirement is 40% and above). I asked why change of decision now and not in April, Dr. Marzo told me that new management of Envoy medical is following strict guidelines on who can be a candidate for esteem and if he still implant the esteem it would be classified as off label device means no support like testing and warranties from envoy medical. I called Envoy and talked to Ploua and she says doctorss makes the final decisions and not Envoy on who should be the candidate esteem and here Dr. Marzo is saying he can do it but Envoy wont let him. I m really confused and frustrated. what should I do?
ReplyDeleteI live in Minneapolis I had the device implanted in my right ear in Feb. 2012. They turned it on in April. It would start to work and then quit. I went back to Chicago four times to see Dr. Marzo to find out why it I was having trouble. In Oct. Dr. Marzo operated on my ear to see if he could correct it. He said it wouldn't work for me . I use a hearing aid in my right ear now but don' hear as well out of right ear as I did before operation.
DeleteBe careful have it looked at by another Dr.
DeleteAnonymous in Chicago area. Just had the August 22 implant done and have many weeks of healing before being activated. I know all the catch 22's your dealing with. Motor vehicals isn't even this bad. Now, you said you have profound loss in those high frequencies but this device only treats loss up to severe loss. There's also other types of hearing loss besides sensorineural like mine which could disqualify you from surgery. Get second opinion please. If Envoy is not supporting you and your Doctor say's this device will be an off the shelf deal? May be Envoy won't send they're engineer to make sure those drivers are put on straight with laser measuring devices. My Doctor just today told me that Envoy will call me when I'm finished healing then call for an activation appoinment, so in your case may be they won't. If anything I learned that you have to be your own advocate, so try other Doctors. Other semi implant devices are available. I Don't know a thing about them though.Also the money issue. Let's face reality here, thousands of people in need of hearing care and they can't afford hearing aids never mind this heart ache. Yeah I know about the "new management" (shudders)deal. Just another way of saying how can we screw 'em today. Good luck freind
ReplyDeleteThnx for imparting the topic. It's so opportune as I am experiencing problems like this official site, and also I am really considering trying this myself.
ReplyDeleteI am awaiting approval to get these implants.... As I figured it, it will cost me $10,280.00 in batteries for the next 40 years for both hearing aids (changed frequently due to my profound loss). Then every 4 years I'd spend $4000 to get new hearing aids, which will cost me $40,000. I need new ear molds every year, which I currently spend $135.00 per ear, which is around $10,800.00. Total is $61,080. The additional benefit that allows me to swim and hear will solely convince me.
ReplyDeleteNow, for the question I have... I've researched it and had an audiogram done recently, I was told by my audiologist that I wouldn't qualify because it's a non conductive loss. Hopefully she is wrong! Will post update once I find out.
I had the implant feb 012 and it is a nightmare for me. Due to infection, they removed it during revison surgery and didn't even install the prosthesis back so I don't even have my old hearing back. Also, no follow-up by Dr. or Envoy. They have my 30,000 now as well as they destroyed my fair hearing in the ear.
ReplyDeletethat is sad to hear.hopefully they will put in the prosthetic so you can go about your business.Thats why i havent done it yet-scared about infection since i have had a kidney transplant
ReplyDeleteI looked here when I needed a hearing aid repair.
ReplyDeleteI had the implant in November 2012. I developed a lot of scar tissue and Dr. Nichols in Houston did a revision in June 2013. It seems to be working pretty good, but just curious if anyone else is experiencing a "bubbling" sound in your ear? Its hard to describe. It's kind-of a rapid vibration. I mentioned it once to Dr. Nichols and I think he said it's the eustachian tube resonating. I think I remember him saying this was normal, but I've asked several people with good hearing if they hear this and they said no. I've tried holding my nose and trying to blow air out my nose and sometimes it helps for a few seconds but comes back. If you've experienced this or heard about this could you please explain what this is. I'm wondering if I still have scar tissue???
ReplyDelete