Recently with all the discussion on catheters and hospital acquired infections, I had the opportunity to conduct an interview with Thomas Cherry from Cook Medical to find out a little bit more on the topic. As mentioned in the interview, CMS is imposing some new rules starting in October that pretty much everyone in the hospital business is aware of, called the 8 conditions that are avoidable, and will not compensate if it is determined the problem lies in one of these areas. Do a quick search of the blog for additional information if needed. Additional product information can be viewed from the Cook website. As is stated below, bloodstream infections is one of the most preventable infections with the use of an impregnated catheter. BD
Email Interview with Thomas Cherry, RN, BSN
Thomas Cherry is the Clinical Product Manager of the Critical Care Division of Cook Medical. Thomas is a Registered Nurse and holds a Bachelors of Science in Nursing from Southeastern Louisiana University, along with certifications in critical care nursing, while also being an active member in professional societies such as APIC (Association for Professionals in Infection Control), SHEA (Society of Healthcare Epidemiology of America), and SCCM (Society of Critical Care Medicine). 
Thank you for taking time out today. The readers on my blog are everyone from high school students, seniors, and healthcare professionals, so first of all could you tell us a bit about catheter-related bloodstream infections (CRBSIs), as many not be familiar with the term and what it is?
First of all, let me discuss it at a higher level and bring it down to CRBSIs, as they are considered one of the most common hospital-acquired infections (HAI). HAIs are infections that are acquired as secondary conditions during a patient’s stay at a healthcare facility. It is important to note these infections are unrelated to a patient’s primary condition.
HAIs are among the most common adverse events in health care, with the Centers for Disease Control and Prevention (CDC) estimating that there are approximately 1.7 million HAIs acquired and 99,000 associated deaths in hospitals each year in the U.S. alone. CRBSIs develop when bacteria enters a patient’s bloodstream through the channel created by a central venous catheter (CVC). It is one of the most preventable infections and it can eventually lead to sepsis, multi-system organ failure and ultimately death. These infections are also very expensive to treat, with treatment costs around $34,000 - $56,000 per infection episode.
The fight against CRBSIs involves multifaceted approaches, including:
· Continuous staff education and training of healthcare personnel,
· Application of maximum sterile barriers during insertion,
· Proper hand hygiene practice,
· Process control measures during insertion (i.e. checklist),
· Utilization of advanced technologies (2 percent CHG skin antisepsis, antimicrobial-catheters, etc.),
· Maintaining proper insertion site care, and;
· Removing unnecessary catheter, when not required.
What types of antibiotics are catheters treated with?
Minocycline and rifampin (M/R) are the two antibiotics impregnated in the Cook Spectrum CVC. These 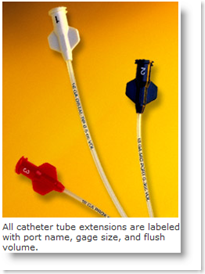 antibiotics were specifically chosen because of their ability to fight methicillin-sensitive and resistant staphylococci (MRSA), as well as both gram positive and gram negative bacteria.
antibiotics were specifically chosen because of their ability to fight methicillin-sensitive and resistant staphylococci (MRSA), as well as both gram positive and gram negative bacteria.
A proprietary process provides antibiotic coverage on both the external and internal surfaces of the catheter to reduce the rates of bacteria colonization on the catheter surface, which reduces the chances of this bacteria entering a patient’s bloodstream.
What motivated Cook to develop an antibiotic-impregnated catheter? Why was this?
While HAIs can develop from a variety of sources, including contaminated bed sheets and dirty hands, it is critical to note more than half of HAIs are attributed to medical devices like CVCs, bladder catheters, endotracheal tubes, tracheostomy devices and surgical implants. Often times, the infection-causing bacteria reside on the patient’s skin surrounding device insertion sites.
The bacteria can attach to the indwelling devices, form antibiotic-resistant biofilms and cause life-threatening infections when inside the body. Essentially, these devices bypass the body’s natural lines of defense against pathogens and provide easy routes for infections to grow. Once this occurs, infections vary from risk level to type based on the host, the medical device and the virulence of the infectious organism.
Considering the high estimated usage rates of CVCs, approximately 3 - 5 million inserted in the U.S. alone with approximately 250,000 - 500,000 episodes of CRBSIs occurring each year, and the limited resources medical professionals have to minimize device-related infections, Cook saw an opportunity to optimize patient safety and catheter technology.
Before creating M/R-CVCs, the team thoroughly reviewed the literature on the best catheter technologies available at that time, such as surface modification technologies and surface coatings engineered to release bactericidal agents in a controlled manner. After a great deal of intensive study conducted at two leading academic research centers (University of Texas/MD Anderson Cancer Center and Baylor College of Medicine in Houston), they discovered it was much more effective to impregnate catheters with antibiotics, specifically minocycline and rifampin, to reduce the risk of CRBSIs.
Is there recent data available about the efficacy of M/R-CVCs?
Yes, the most recent research was presented by Dr. Issam Raad at the 18th annual Society for Healthcare Epidemiology of America demonstrating that the M/R-impregnated CVCs reduced the risk of bacteremia and also the likelihood that bacteria could become resistant to the antibiotics used to prevent their colonization. Dr. Raad showed that this combination of drugs actually reduces the risk of bacteria developing antibiotic resistance to the drugs used in the MR-CVCs.
It was previously thought by some medical researchers that the use of these devices could promote antimicrobial resistance, as conventional catheters are not impregnated with antibiotics and can more easily allow bacteria to colonize and spread to the patient’s blood stream. However, this recent evidence suggests otherwise. 
Are M/R-impregnated catheters more expensive than standard catheters?
M/R-CVCs are more expensive than standard catheters. Nevertheless, as mentioned previously, treating CRBSIs after they occur can create a greater financial burden on health care facilities. Additionally, catheters are typically sold as ‘sets’ or ‘trays.’ A set may include the basics a physician would need to place the catheter, including a needle, syringe, guide wire, scalpel, and catheter, whereas a tray would include all that is in a set plus any dressing, drapes, drugs or fluids required. A Cook tray with M/R that is used in the majority of CVC placements may cost about $125. A similar non-coated catheter tray from a competitor may cost $65.
(Not much of a price difference if a human life is potentially at stake...BD)
What are your feelings on how the M/R-CVC can prevent the incidence of CRBSIs in the future and how can we better educate the public?
M/R-CVCs are a proven technology and I believe that to effectively reduce the occurrence of CRBSIs hospitals must utilize the most effective prevention processes, as well as evidence-based medical device technologies. Hospital employees understand that proper hygiene and prevention protocol, such as the utilization of checklists, dramatically reduce the rates of the HAIs. Nevertheless, Americans still are contracting HAIs and many are dying. Process control measures, increased education or advance technologies cannot be implemented singularly; all three strategies enacted together will work together to prevent these infections. A complete comprehensive approach working in conjunction will be the only approach that will help achieve the goal of zero tolerance.
It’s apparent that other strategies are needed to successfully lower the occurrence rates of HAIs. We use technology everyday to improve the quality of our personal and professional lives and its only natural we look towards technology to promote positive outcomes in healthcare.
In the coming months, I expect there will be a great deal of media attention around HAIs with the formal enactment of the Centers for Medicare & Medicaid Services (CMS) reimbursement policy change, whereby as of Oct. 1, 2008 CMS will no longer reimburse hospitals for additional treatment costs of patients with eight conditions it deems avoidable, including CRBSIs. This, coupled with the sponsorship of mandatory infection reporting in over 22 states, will serve to cast more light on HAIs and inform the public of the risks once acquired and the challenges in treatment options.
Additionally, a public health campaign sponsored by the Center for Disease Control and Prevention would go far in raising the awareness around the seriousness of the issue and empower the public to become better health consumers.
End of Interview…and thanks to Thomas Cherry for the time and information...



0 comments :
Post a Comment