The idea here is to provide continuous blood glucose data day and night and as most wireless devices are being made today, improve patient compliance This is only stage one so it will be a while before any FDA approval is given. BD
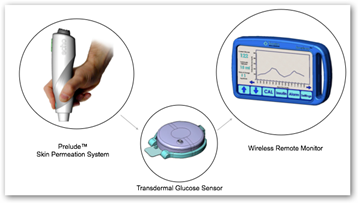
FRANKLIN, Mass., April 27 /PRNewswire-FirstCall/ -- Echo Therapeutics, Inc. (OTC Bulletin Board: ECTE), a company developing its needle-free Symphony™ tCGM System as a non-invasive, wireless, transdermal continuous glucose monitoring (tCGM) system and its Prelude™ SkinPrep System for transdermal drug delivery, in collaboration with its strategic partner, Ferndale Pharma Group, Inc., announced today that the first patients were enrolled in a clinical study of its Prelude SkinPrep System.
This clinical study is designed to evaluate the ability of the Prelude SkinPrep System to ablate the skin prior to application of OTC 4% lidocaine cream for local dermal anesthesia. Upon completion of this study, Ferndale and Echo anticipate submitting a 510(k) premarket notification to the United States Food and Drug Administration (FDA) with subsequent commercial launch of the product after 510(k) clearance.



This tool is an innovation which is very useful in helping people with diabetes. I hope this tool immediately pass the test in a clinical trial
ReplyDeleteGod me too,coming from someone who suffers from needle phobia i WANT this!!!!!!!!!
ReplyDelete