The device, called the LINX Reflux Management System, is being studied at UC San Diego Medical Center as part of a US and European multicenter clinical trial. Today Europe has announced their approval for the device. The link below gives some additional details from my post last year.
Implanted Medical Device for GERD (Gastroesophageal Reflux Disease) in Trials
In many areas devices are competing with drugs and with this 20-30 minute procedure and successes it is having, we may have another winner in the device versus drug competition. BD
Magnetic Device Studied As Treatment For Heartburn And Acid Reflux
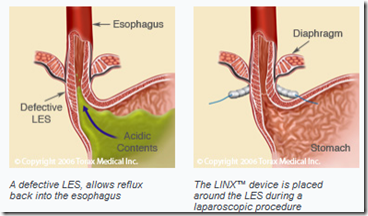
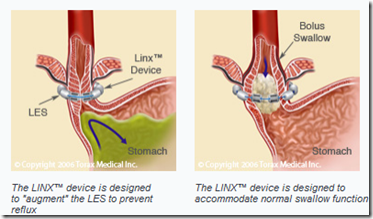
Torax Medical Inc. won CE Mark approval in the European Union for an implantable device that treats patients suffering from acid-reflux disease.
The company’s Linx device is intended for patients with gastro-esophageal reflux disease, which occurs when stomach juices flowing upward into the esophagus. The disease is caused by defects in the esophageal sphincter muscle, which typically prevents acid and bile from entering the esophagus.
Torax Medical wins CE Mark for GERD device | MassDevice - Medical Device Industry News



GERD symptoms is a big problem it should be cures as fast as possible
ReplyDelete