Back in March, DePuy sent a letter to doctors advising that the ASR was being phased out. The woman filing the lawsuit had problems, pain, and the device failed and was replaced with a device from another company. The suit alleges that the company knew of the defects after receiving FDA approval but doctors were not warned. 
DePuy (Johnson and Johnson) Warning Doctors of High Failure Rates – Hip Product Being Phased Out
As the article mentions here, there could be more legal suits arising and Johnson and Johnson bought another knee/hip replacement company in December of 2009.
Johnson and Johnson Company, DePuy Orthopedics Acquires Finsbury Orthopaedics from the UK
When having to have a replacement knee or hip or any medical device replaced and under go surgery again, there’s always the risk for infection exposure as you don’t want MRSA in your bone areas at all, and that in itself has been another cause of hip/knee replacements to be done over. BD
DePuy Orthopaedics is facing the first of what might be an avalanche of lawsuits over one of its hp implants, after a Florida woman sued the company over an allegedly faulty hip implant.
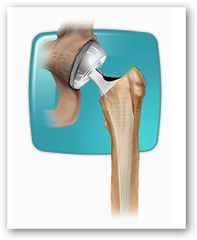
Kathleen Margenau was implanted with a DePuy ASR acetabular cup in March 2008, according to court documents. Following the surgery, Margenau began experiencing pain and "extreme weakness in her hip and quadriceps," according to the lawsuit. Little more than a year later, her surgeon explanted the device and found that the device had "catastrophically failed." Another device made by one of the Johnson & Johnson (NYSE:JNJ) subsidiary's competitors was implanted and Margenau returned to her active lifestyle, according to the document.
Kathleen Margenau was implanted with a DePuy ASR acetabular cup in March 2008, according to court documents. Following the surgery, Margenau began experiencing pain and "extreme weakness in her hip and quadriceps," according to the lawsuit. Little more than a year later, her surgeon explanted the device and found that the device had "catastrophically failed." Another device made by one of the Johnson & Johnson (NYSE:JNJ) subsidiary's competitors was implanted and Margenau returned to her active lifestyle, according to the document.
DePuy discontinued the cup and had phased it out by March of this year, sending a letter to doctors that month warning that data from the Australian medical device registry showed a "higher-than-expected" failure rate in traditional hip replacements, especially in smaller patients or those with weak bones.
Florida woman sues DePuy Orthopedics over hip implant | MassDevice - Medical Device Industry News




0 comments :
Post a Comment