The trial will be carried out at several locations and Inspire is a spinoff of Medtronic.  The constant interruption of sleep can lead to cardiac issues. On each breath the nerve in your neck restores muscle tone to the tongue and it does not collapse. I have seen some of the sleep at home units where you put on a headset at night and this does look a little better than that, but there’s the object inside your body with an implant to think about. I first wrote about this device about a year or so ago.
The constant interruption of sleep can lead to cardiac issues. On each breath the nerve in your neck restores muscle tone to the tongue and it does not collapse. I have seen some of the sleep at home units where you put on a headset at night and this does look a little better than that, but there’s the object inside your body with an implant to think about. I first wrote about this device about a year or so ago.
Implant to Treat Obstructive Sleep Apnea – Inspire Medical Systems
Here’s another link on a sleep at home system for comparison.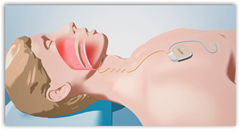
Sleep Apnea Testing Done at Home – Less Expensive Than Sleep Clinics Solution
Germany, Belgium and The Netherlands have already begin with the Star trial program and you can find more information here on contacting the company regarding the trial or the implant. BD
Inspire Medical Systems, a leading developer of neuro-stimulation therapies for the treatment of obstructive sleep apnea (OSA), announced today that the company has received approval from the U.S. Food and Drug Administration (FDA) to begin its STAR pivotal clinical trial. The STAR trial (Stimulation Therapy for Apnea Reduction), is a multi-center study that will evaluate both the safety and effectiveness of Inspire™ Upper Airway Stimulation (UAS) therapy in patients with moderate to severe obstructive sleep apnea (OSA). The STAR trial will be conducted at leading medical centers across the United States and Europe. The results of this study will be the basis for a Pre-market approval (PMA) application to the FDA.
Leading medical centers in Germany, Belgium and The Netherlands are currently approved to implant patients into the STAR trial. Professor Paul Van de Heyning, Director of Otolaryngology and Head and Neck Surgery at the University of Antwerp in Antwerp Belgium recently implanted the first patient in the STAR trial
Inspire Medical Systems Announces FDA approval for Pivotal Clinical Trial | Benzinga.com




I am part of the Inspire study and had this implanted sept 2011. It works great!!! No apnea during follow up study. no more CPAP. can roll around my bed now. no lugging that stupid machine around. Very non intrusive. I couldn't wait 5 or 7 years for it to come to market so I join the study. For those apnea suffers who can't stand the CPAP, your savoir will come. It will just be a few more years.
ReplyDelete