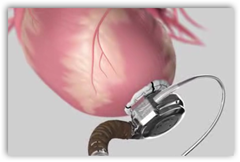
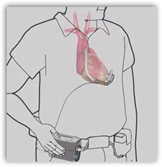
The tiny pump is the size of a golf ball and is attached to the heart and a wire comes out through the skin to an externally worn controller. The device is a heart pump for  those who basically have no other alternatives, or could be waiting for a transplant possibly. Two lithium batteries run the unit. The controller and batters are contained in a case and worn either around the neck or over the chest. The device is approved for those with late stage heart failure and is truly a life saver. Back in June of 2009 the first 3 patients to receive the pump were in New York.
those who basically have no other alternatives, or could be waiting for a transplant possibly. Two lithium batteries run the unit. The controller and batters are contained in a case and worn either around the neck or over the chest. The device is approved for those with late stage heart failure and is truly a life saver. Back in June of 2009 the first 3 patients to receive the pump were in New York.
First heart patients implanted with 3rd Generation mechanical heart pump – DuraHeart
The device is a left-ventricular assist device, or LVAD and the first devices came into play in the 80s actually. BD
FRAMINGHAM, Mass. and SYDNEY, Jan. 24, 2011 /PRNewswire/ -- HeartWare International, Inc. (Nasdaq: HTWR; ASX: HIN), a leading innovator of less invasive, miniaturized circulatory support technologies that are revolutionizing the treatment of advanced heart failure, today announced that the U.S. Food and Drug Administration (FDA) has approved an IDE (Investigational Device Exemption) Supplement that allows HeartWare to enroll a third allotment, of 94 additional patients, in
its "ADVANCE" bridge-to-transplant clinical trial under a Continued Access Protocol (CAP). The FDA granted two prior allotments of 54 patients each in April and September 2010, and the final patient from the second cohort was implanted earlier this month.
HeartWare's ADVANCE clinical trial is an FDA approved IDE study
designed to evaluate the HeartWare® Ventricular Assist System as a bridge to heart transplantation for patients with end-stage heart failure. Under the ADVANCE study, 140 patients at 30 U.S. clinical sites received HeartWare HVAD® pumps, making it the largest bridge-to-transplant pivotal trial to date. HeartWare submitted to the FDA a Pre-Market Approval (PMA) application seeking approval of the HeartWare System for the bridge-to-transplant indication in December 2010.
Related Reading:



0 comments :
Post a Comment